Recall: The label says muscle relaxer, but arthritis drugs are in the bottle
Unichem Pharmaceuticals has recalled one lot of a muscle relaxer because the pills in the bottle might not be the muscle relaxer on the label.
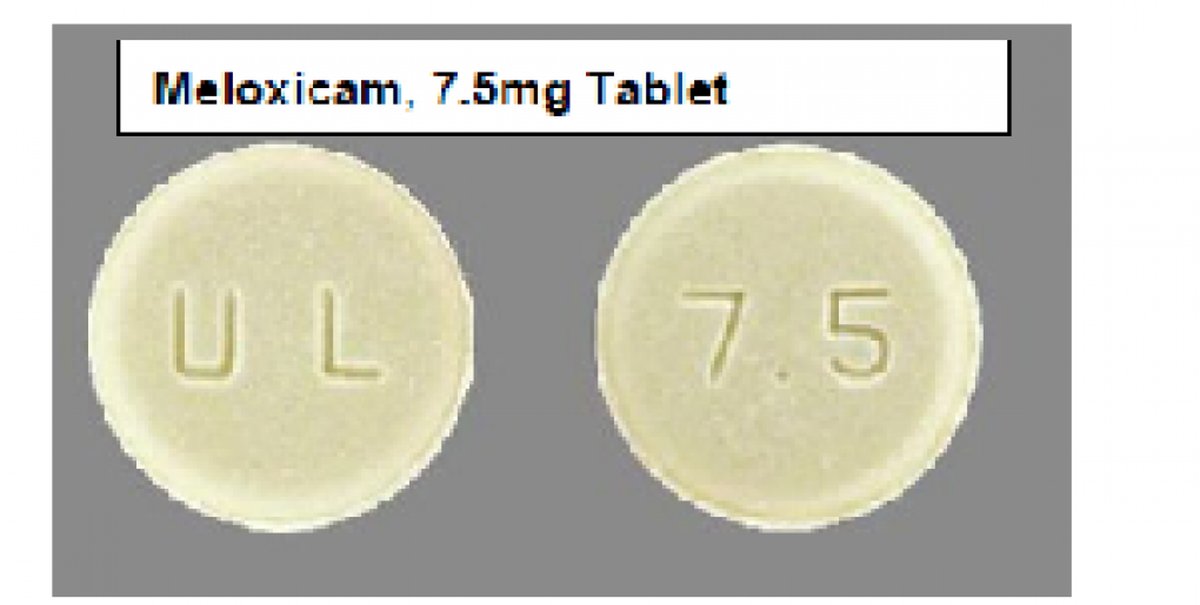
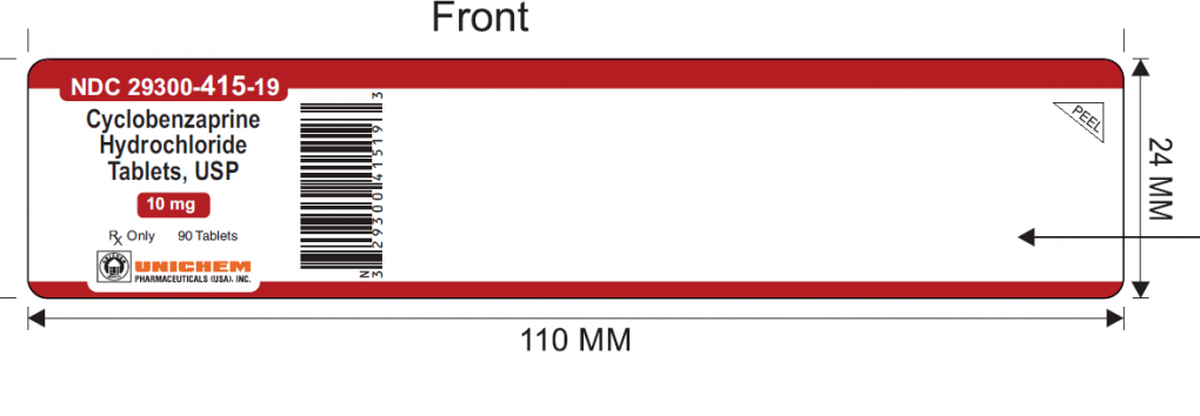
The East Brunswick, New Jersey, company’s FDA-posted recall notice says the label for 90-count bottles of Cyclobenzaprine Hydrochloride 10 mg tablets got put on at least one bottle of Meloxicam 7.5 mg tablets. Meloxicam is a non-steroidal anti-inflammatory drug that treats various forms of arthritis.
“For patients who unknowingly take Meloxicam there is a reasonable probability of serious adverse events, including cardiovascular, gastrointestinal, renal, anaphylaxis, and skin reactions,” the recall risk statement says.
This is especially the case for people taking associated blood thinners or anti-inflammatory drugs, people allergic to Meloxicam or people with a pre-existing medical condition.

Cyclobenzaprine tablets are blue with a “12” on one side and a “U” on the other side. Meloxicam tablets are yellow with a “U & L” on one side and “7.5” on the other side. The recalled lot carries No. GMML24026A with an expiration of September 2027.
If you’re a consumer, take the recalled tablets back to the pharmacy where you got them. Direct questions to Inmar, which is handling the recall for Unichem, at 877-840-5109 or rxrecalls@inmar.com, Monday through Friday, 10 a.m. to 6 p.m., Eastern time. Pharmacies are asked to notify consumers and call Inmar for directions on returning the recalled tablets.
Take any medical problems to a medical professional. Then notify the FDA via the MedWatch program, either by filling out the form online or calling 800-332-1088 to get a form, then submitting it by mailing it to the address on the form or faxing it to 800-FDA-0178.
This story was originally published September 2, 2025 at 3:06 PM with the headline "Recall: The label says muscle relaxer, but arthritis drugs are in the bottle."
