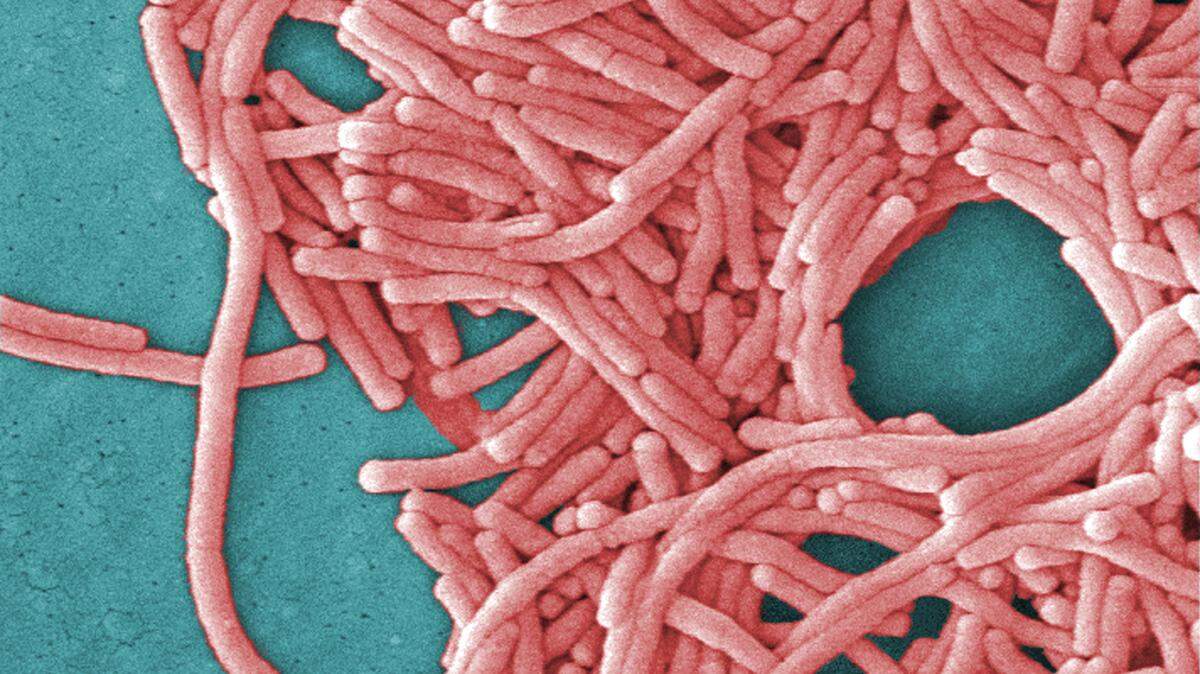
Legionella is scary. It lurks in water and can kill or sicken us. Here is what we can do
READ MORE
The threat of Legionella | The State Worker
California state workers returning to offices after working remotely during the COVID-19 pandemic could face heightened risks of contracting another dangerous respiratory infection: Legionnaires’ disease.
Expand All
Imagine this scenario: Hospitals in one part of the world report a rapidly growing number of patients showing up with “atypical” pneumonia that cannot be assigned to any known cause.
Scary, right?
This scenario has unfortunately played out many times throughout recent history. In 2002, it was the first cases of Severe Acute Respiratory Syndrome, or SARS. It happened again in 2012 with Middle East Respiratory Syndrome, or MERS. Then, of course, late 2019 saw the emergence of COVID-19.
Although as a society we tend to forget such events with relative speed, I vividly remember the frightening scenario that played out in the summer of 1976, when I was 8 years old and living in Bethesda, Md. News surfaced of a strange pneumonia of a then-unknown cause that was killing people in nearby Pennsylvania.
Many were showing up to hospitals in Pennsylvania (and a few other locations) with the atypical pneumonia. Many were very ill, and some died. Although the disease’s origins were unclear, there was one key clue: 100 patients with this pneumonia had spent time in Philadelphia during a conference of the Pennsylvania American Legion.
This caused panic around the country and in my area. The fear was palpable — particularly for me, as I was living near the National Institutes of Health (where both my parents worked) and surrounded by people focused on and infectious diseases.
Extensive research eventually determined the cause of the pneumonia to be a previously unknown bacterium now named “Legionella pneumophila” . The resulting illness would become known as Legionnaires’ disease.
The spread of the bacteria was ultimately traced to an unusual source for such a pathogen: It lurked in the water of the air conditioning system of the hotel that hosted the convention.
Since then, society has learned a lot about Legionnaires’ disease and the bacterium that causes it.
On a positive note, the disease is treatable, especially if diagnosed early, with antibiotics. Unfortunately, however, Legionella and its close relatives have been associated with a growing number of cases per year.
More than 10,000 cases were reported in 2018, and the actual number of cases was probably higher. High-profile outbreaks unfolded in 2014 and 2015 in Flint, Mich.; in New York City in 2015; and at Disneyland in 2017.
The exact reasons for the rising cases are not known, but they tend to share a feature that is of great importance in controlling this disease. Investigations of Legionnaires’ outbreaks invariably point to contaminated water, usually stagnant, in water-bearing systems such as hot tubs, showerheads, sinks, fountains, hot water tanks, or cooling towers.
Though the possibility that deadly pathogens lurk in our water is certainly scary, it also means one can prevent the pathogen from infecting people in many cases by assessing, modifying, and cleaning water storage and distribution systems.
When new Legionnaires’ cases arise, it’s critically important to figure out where the bacteria was lurking and how it spread to people. A key component in such work these days involves what is known as genomic epidemiology, in which one determines the genomic content of the bacteria from patients and environmental samples and then builds “family trees” to identify its likely sources.
This is akin to human ancestry tests and crime scene DNA collection but with a focus on the bacteria and its DNA. Once one finds the likely source of the bacteria in infected people, one can prevent future infections from that source by removing, cleaning, flushing, or modifying the water system where it was found.
Important preventive measures can also be taken before any outbreak has occurred. Just as we try to eliminate standing water to prevent mosquito breeding, we can assess water systems in buildings and other facilities for the presence of the bacteria before it causes disease. Unfortunately, such measures are not always pursued rigorously enough.
And as many water systems have been stagnating during the COVID19 pandemic, addressing Legionella risks is particularly important now.
Outbreaks of Legionnaires’ are an important reminder that there are infectious diseases other than COVID-19 that we need to keep our eyes on. Alas, many of us, myself included, have been lost in the world of the coronavirus. Over the last 18 months, in my role as a professor and microbiologist at UC Davis, I have spent most of my time working on the genomics, genomic epidemiology and transmission of Sars-CoV-2 (the virus that causes COVID-19) as well as teaching and communicating about the pandemic.
However, I do have memories of working on the genomics and genomic epidemiology of hundreds of different microbes before this one. This has included many that do not cause any diseases (my favorites, actually) as well as the causes of AIDS, flu, tuberculosis, malaria, cholera, and anthrax. It also included some work on Legionnaires’ disease: My lab at UC Davis helped carry out DNA analysis that was used for the genomic epidemiology of samples from the Flint outbreak.
Though it might be nice to forget about all the other infectious diseases, we should not. We need to remember to keep working to predict, prevent and control their spread and effects, too. That is what I plan to keep doing during and certainly after the COVID-19 pandemic.
Jonathan Eisen is Professor in Medical Microbiology and Immunology, Evolution and Ecology, and the Genome Center at UC Davis
This story was originally published September 22, 2021 at 5:00 AM.